In the course of developing a historical database of British cement chemistry, it has been necessary to characterise all the raw materials that have been used historically. Many raw materials are sufficiently homogenous that a few typical sample analyses are sufficient. Pure Chalk, in particular, needs little data other than some generalised information on minor and trace elements. However, the Lower Chalk is much more problematic because of its vertical and lateral variability.
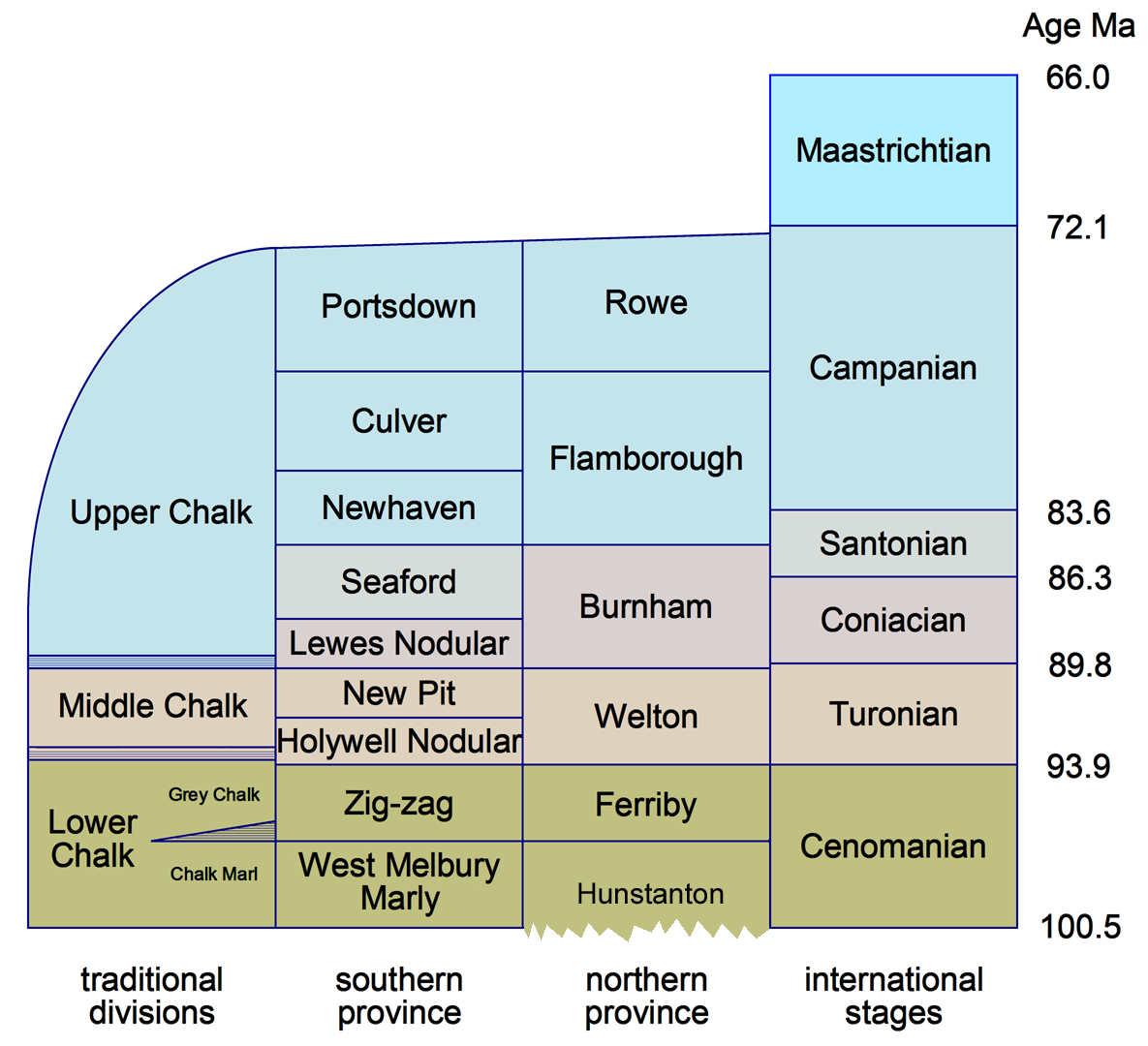
The Lower Chalk (as defined by nineteenth century geologists) consists essentially of the Cenomanian age of the Upper Cretaceous. The succeeding five ages (Turonian, Coniacian, Santonian, Campanian and Maastrichtian) are represented by pure chalks.
The Cenomanian age began 100.5 million years ago and ended 93.9 million years ago. It gets its name, as do most of the Cretaceous ages, from a type location in France - in this case Le Mans (the Celtic Cenomani tribe lived in the district). It varies significantly across England, which is conventionally divided into a Northern and Southern province, with, perhaps, a transitional zone in between. In both provinces it is divided into two Formations. In the Southern Province, the West Melbury Marly Chalk forms the lower division and the Zig-zag Chalk the upper. These formations correspond closely with the Chalk Marl and Grey Chalk of nineteenth century geology. In the Northern Province, the Ferriby Chalk corresponds closely to the Zig-zag Chalk of the south, but the lower half of the stage shrinks almost to nothing; instead the Hunstanton Formation consists of the thin Red Chalk (which spans the Albian and Lower Cenomanian). This thinning of the Lower Cenomanian is the chief distinguishing characteristic of the two provinces. An exact transition from Chalk Marl and Gault to Red Chalk is not visible, because the boundary is buried beneath fenland.
The Chiltern Hills constitute the chalk escarpment running from Wallingford on the Thames north-eastwards through Oxfordshire, Buckinghamshire, Hertfordshire and Bedfordshire until it disappears under the Cambridgeshire fens. Seventeen cement plants listed in this website were strung along the escarpment and extracted their raw materials from it. Although these raw materials have a lot in common, there are distinct trends in chemistry on passing along the escarpment. In particular, the escarpment spans the boundary between the Southern and "transitional" provinces. The trend can be exemplified by plotting the carbonate-content profiles of the stratigraphic column at various sites.
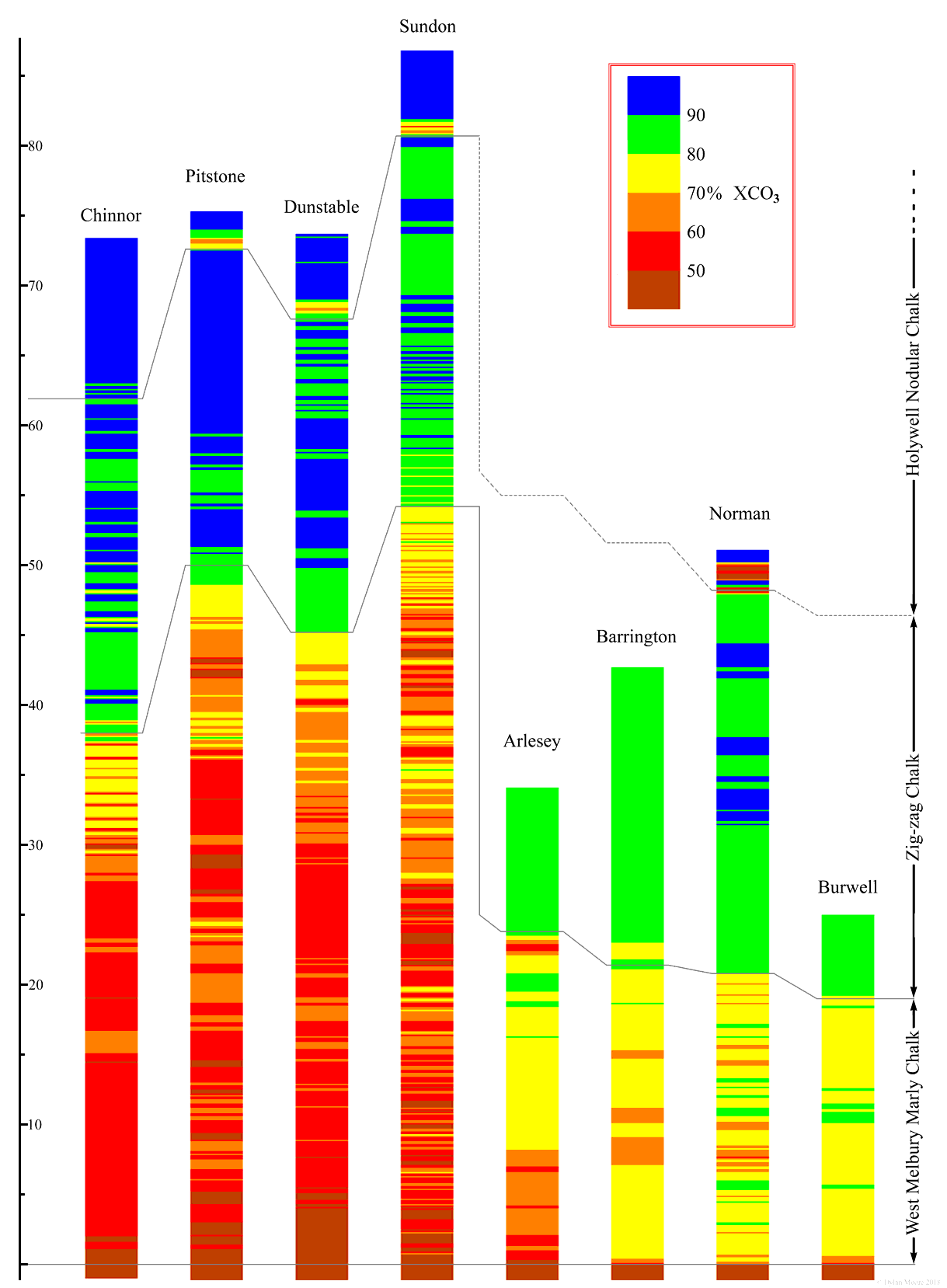
The chart shows the thickness (in metres) of the Cenomanian and its carbonate content. Each column is a composite of a large number of correlated data sources which might be boreholes, trenches or surface samples; none had a single borehole passing through all levels. The gathering of this data began for me back in the mid-1970s, when I gathered large numbers of samples from exactly identified horizons in Sundon quarry. Colourimetric analysis of these on free weekends ceased when the plant was shut down. Hundreds of samples were subsequently analysed on the Humber Telsec TXRF, and I finally understood the intricacies of Sundon quarry some months after the plant closed. Story of my life.
Carbonate content is the most obvious variable characteristic of chalk, and reflects the balance between "pure" carbonate-based fossil fragments and "normal" sediments delivered from rivers that are normally silty or clay-like, bearing silica, alumina, iron oxides and many other minor elements. It is necessary to discuss what exactly is meant by "carbonate content". It is a parameter that has been used fairly indiscriminately in the cement industry ever since it started (in the 1860s, in Germany) to become "scientific". Early practitioners measured it with an instrument mis-named "calcimeter", the name of which expresses the confusion from the outset, since it measures not calcium, but carbon dioxide. In early texts, the term carbonate meant "CO2 expressed as CaCO3".
Partly from the influence of German workers, who were familiar with limestones containing dolomite, it became apparent that CO2 content might be associated with magnesium as well as calcium, and this led to the assumption that all the calcium and all the magnesium in a rawmix are present as carbonates. Many published rawmix analyses right up to the end of the nineteenth century were based on this assumption. However, in the case of British chalks (Note 1), this assumption is entirely incorrect: British chalk contains little or no dolomite except in a few exposed coastal locations, and magnesia in chalk-region rawmixes derives almost entirely from the argillaceous component, where it exists as silicates, micas, etc. (Note 2) Furthermore, not all calcium exists as carbonate. Nearly all the phosphorus in chalk exists in the form of apatites, which tie up 1.32 g of CaO for each 1 g of P2O5. Phosphates are particularly significant in the Lower Chalk. Later in the twentieth century, with element-specific analysis methods, "calcium carbonate content" was redefined as "CaO expressed as CaCO3", again introducing significant error. I can't imagine that any consumers of this data ever questioned it. In this work, carbonate content means "total mass % of XCO3" where X represents all cations actually present in the carbonate phases.
The chart shows a number of obvious characteristics of the Lower Chalk.
- The Zig-zag Chalk, although impure, has higher carbonate in general than the West Melbury Chalk
- Both formations are variegated with hundreds of marl/chalk alternations.
- The West Melbury Marly Chalk reaches its maximum thickness at Sundon, but further north-east gets much thinner.
The change between Sundon and Arlesey (only 17 km apart) is particularly large, and may in fact be abrupt, associated with the "Lilley Bottom Structure", which is inferred from gravitational anomaly data to be the north-eastern edge of a major basement structure. This dividing line has other effects:
- The lowest part of the West Melbury formation takes the form of the Glauconitic Marl Member south-west of this line - to the north-east it becomes the phosphate-yielding Cambridge Greensand Member.
- The uppermost part of the Albian, below the Cenomanian, takes the form of Upper Greensand to the south-west of this line. Borehole data on the extent of the Upper Greensand under the London Basin shows that this boundary extends to the south-east as far as Maidstone.
To the southwest of the Lilley Bottom boundary, the north-eastward increasing trend in West Melbury thickness is interrupted at Dunstable, where in the vicinity of Totternhoe, the uppermost part of the formation is missing.
The use of the Chalk Marl for cement in the region had its origins in the phosphate industry. To the northeast of the Lilley Bottom boundary, porous Cambridge Greensand overlying impervious Gault clay leads to a line of springs along the contact of the outcrop. These were known locally as "fossil springs" because of the phosphatised fossils that wash out of the Greensand at these sites. The base of the Cenomanian was therefore unusually well known long before formal geology was investigated. When the phosphate industry began in the mid-nineteenth century, local landowners dug along the spring-line to extract the phosphatic nodules. This led to Chalk Marl being side-cast and landowners looked for a use for it. All the earlier cement plants were originally phosphate-digging sites. The approximate chronology is:
- 1883: Arlesey
- 1884: Standard
- 1888: Romsey Town
- 1891: Cam Blue Lias
- 1892: Burwell
- 1892: East Anglian
- 1893: Rhee Valley
- 1895: Hauxton
- 1896: Bottisham Lode
- 1898: Meldreth
- 1901: Saxon
- 1904: Norman
- 1927: Barrington
South-west of Lilley Bottom, the four plants began later:
The latter plants were sited on radiating rail routes and were intended to exploit the Lower Chalk (offering cheap cement manufacture) to supply the London market. All these benefitted from a number of the characteristics of the raw material not available elsewhere (Note 3):
- a highly siliceous rawmix, but with the silica in ultrafine form, allowing the production of cements high in silicates, therefore providing high concrete durability
- a highly siliceous clinker, requiring low grinding energy
- materials mainly close to kiln feed chemistry, so that minimal blending was required
- simple, one-pit quarrying operations that led to low material costs
All this conspired to make (at least potentially) for very low manufacturing costs. Sundon, in particular, was the lowest-cost British cement manufacturing plant for more than half its 77 year existence.